Skyrizi Brochure
Skyrizi Brochure - Skyrizi is used to treat patients 16 years and older with moderate to severe crohn’s disease and adults with moderate to severe ulcerative. Skyrizi is available in a 600 mg/10 ml intravenous infusion and a 360 mg/2.4 ml. See important safety information and prescribing. See the detailed “instructions for use” that comes with skyrizi for information on how to prepare and inject a dose of skyrizi, and how to properly throw away. Skyrizi (risankizumab injection) is indicated for the treatment of adult patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy. It will also help you understand. This brochure will help you better understand skyrizi and the reasons your doctor may have prescribed it for you, as well as important safety information. Skyrizi is a treatment for moderate to severe crohn’s disease that works diferently. Dispense as written/brand medically necessary substitution permitted kg. Skyrizi (risankizumab injection / risankizumab for injection) is indicated for: Patient resourcessee the resultstalk with your doctorpatient videos Skyrizi (risankizumab injection / risankizumab for injection) is indicated for: In this brochure, you’ll learn more about skyrizi as a uc treatment option and if it could be the right fit for you. Skyrizi contains the active substance risankizumab. Stable to week 12 in patients treated with skyrizi in crohn’s disease. Get to know skyrizi, an fda‐approved biologic treatment for adults with ps, psa, cd, & uc. Skyrizi is a prescription medicine used to treat adults: Skyrizi is indicated for the treatment of In this brochure, you’ll learn more about skyrizi as a uc treatment option and if it could be the right fit for you. Skyrizi is used to treat patients 16 years and older with moderate to severe crohn’s disease and adults with moderate to severe ulcerative. These are inflammatory conditions that affect the skin and nails. Skyrizi is used to treat patients 16 years and older with moderate to severe crohn’s disease and adults with moderate to severe ulcerative. Skyrizi is used to treat adults with moderate to severe crohn’s disease, an inflammatory condition affecting the gastrointestinal tract that can cause abdominal pain, severe diarrhea, fatigue,. Stable to week 12 in patients treated with skyrizi in crohn’s disease. Skyrizi is available in a 600 mg/10 ml intravenous infusion and a 360 mg/2.4 ml. It will also help you understand. In this brochure, you’ll learn more about skyrizi as a uc treatment option and if it could be the right fit for you. Skyrizi is used to. Dispense as written/brand medically necessary substitution permitted kg. One of the key proteins responsible for inflammation. See important safety information and prescribing. In this brochure, you’ll learn more about skyrizi as a uc treatment option and if it could be the right fit for you. • with moderate to severe plaque psoriasis who may benefit from taking injections or pills. Skyrizi contains the active substance risankizumab. • with moderate to severe plaque psoriasis who may benefit from taking injections or pills (systemic therapy) or treatment using. You’ll want to aim for relief. • the treatment of adults with moderately to severely active crohn's disease who have had an inadequate. Patient brochure provides patients with an overview of how skyrizi works,. Patient brochure provides patients with an overview of how skyrizi works, including key efficacy, safety, and dosing information. Skyrizi is used to treat patients 16 years and older with moderate to severe crohn’s disease and adults with moderate to severe ulcerative. Skyrizi is indicated for the treatment of moderately to severely active crohn’s disease in adults. Skyrizi is a prescription. Skyrizi is a treatment for moderate to severe crohn’s disease that works diferently. Skyrizi (risankizumab injection / risankizumab for injection) is indicated for: Get to know skyrizi, an fda‐approved biologic treatment for adults with ps, psa, cd, & uc. Skyrizi is used to treat patients 16 years and older with moderate to severe crohn’s disease and adults with moderate to. Skyrizi (risankizumab injection) is indicated for the treatment of adult patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy. In this brochure, you’ll learn more about skyrizi as a uc treatment option and if it could be the right fit for you. Skyrizi is a treatment for moderate to severe crohn’s disease that works. It will also help you understand. In this brochure, you’ll learn more about skyrizi as a uc treatment option and if it could be the right fit for you. • with moderate to severe plaque psoriasis who may benefit from taking injections or pills (systemic therapy) or treatment using. Skyrizi delivers early and significant relief from many ulcerative colitis (uc).. In this brochure, you’ll learn more about skyrizi as a uc treatment option and if it could be the right fit for you. Get to know skyrizi, an fda‐approved biologic treatment for adults with ps, psa, cd, & uc. Skyrizi delivers early and significant relief from many ulcerative colitis (uc). You’ll want to aim for relief. See the detailed “instructions. Dispense as written/brand medically necessary substitution permitted kg. Patient brochure provides patients with an overview of how skyrizi works, including key efficacy, safety, and dosing information. It will also help you understand. One of the key proteins responsible for inflammation. See important safety information and prescribing. Skyrizi contains the active substance risankizumab. • the treatment of adults with moderately to severely active crohn's disease who have had an inadequate. Patient resourcessee the resultstalk with your doctorpatient videos Skyrizi is indicated for the treatment of Dispense as written/brand medically necessary substitution permitted kg. See the detailed “instructions for use” that comes with skyrizi for information on how to prepare and inject a dose of skyrizi, and how to properly throw away. Skyrizi is used to treat patients 16 years and older with moderate to severe crohn’s disease and adults with moderate to severe ulcerative. Skyrizi is used to treat adults with moderate to severe crohn’s disease, an inflammatory condition affecting the gastrointestinal tract that can cause abdominal pain, severe diarrhea, fatigue, and. See important safety information and prescribing. Stable to week 12 in patients treated with skyrizi in crohn’s disease. Skyrizi delivers early and significant relief from many ulcerative colitis (uc). Skyrizi is available in a 600 mg/10 ml intravenous infusion and a 360 mg/2.4 ml. • with moderate to severe plaque psoriasis who may benefit from taking injections or pills (systemic therapy) or treatment using. Skyrizi is indicated for the treatment of moderately to severely active crohn’s disease in adults. Get to know skyrizi, an fda‐approved biologic treatment for adults with ps, psa, cd, & uc. Skyrizi (risankizumab injection / risankizumab for injection) is indicated for:NDC 00074210001 Skyrizi 150 mg/mL Details HelloPharmacist
Crohn’s Disease Resources SKYRIZI® Complete for Crohn’s Disease
Skyrizi Approved for Moderate to Severe Plaque Psoriasis Dermatology
New Drug Product Skyrizi MPR
SKYRIZI® (risankizumabrzaa) for Psoriatic Arthritis
Skyrizi Injection FDA prescribing information, side effects and uses
Understanding Insurance Skyrizi Complete
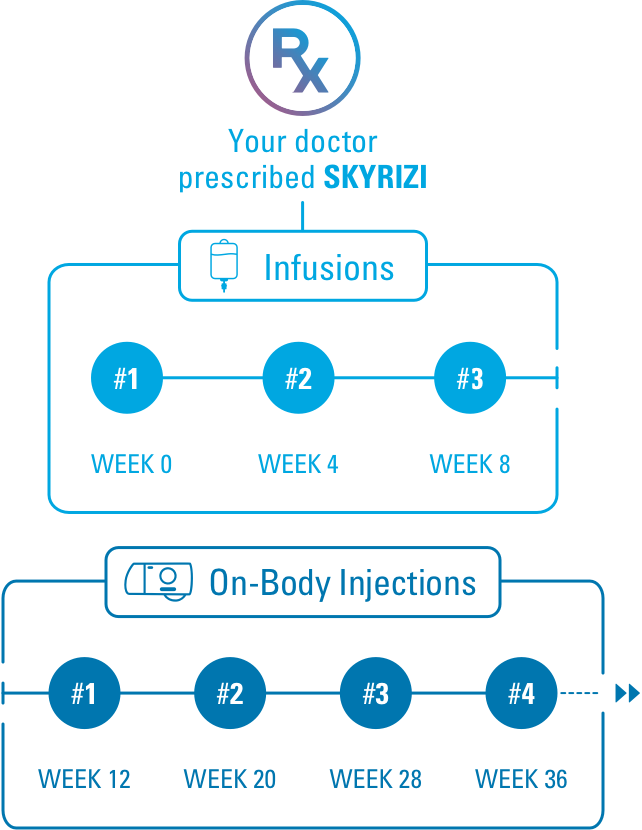
SKYRIZI® (risankizumabrzaa) Dosing for Ulcerative Colitis
SKYRIZI® (risankizumabrzaa) A Biologic Treatment for Psoriasis
New Indication Skyrizi for Crohn Disease MPR
See Full Safety And Prescribing Information.
Skyrizi Is A Prescription Medicine Used To Treat Adults:
This Brochure Will Help You Better Understand Skyrizi And The Reasons Your Doctor May Have Prescribed It For You, As Well As Important Safety Information.
View Resources For Your Practice And Patients, Including Information On Getting Access To Skyrizi, Dosing Information, And Injection Support.
Related Post: